2019-08-27
Inflammatory diseases: Scientists develop new antibody with therapeutic potential
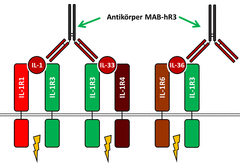
(Click to enlarge) Interleukins IL-1α and IL-1β bind to the IL-1 receptor which consists of the subunits IL-1R1 und IL-1R3 (green). This leads to signal transduction inside the cells triggering pro-inflammatory processes (flash symbol). Accordingly, IL-33 binds to IL-1R4 and IL-R3. IL-36α, IL-36β and IL-36γ bind to IL-1R6 and IL-1R3. The novel antibody MAB-hR3 blocks the shared subunit IL-1-R3 and interrupts the signaling cascade of all six interleukins. (Figure [M]: M. Wegmann, 2019)
In collaboration with an international team, DZL scientists from Research Center Borstel developed and tested a new antibody which might be used against inflammatory diseases. The results of this study were published in Nature Immunology.
Interleukins (IL) of the IL-1 family play a role in many inflammatory diseases, because their receptor system promotes pro-inflammatory effects (see figure). They trigger immune cells to release cytokines responsible for disease pathogenesis. Strategies have been developed to turn off the effect of single interleukins in order to ameliorate symptoms of diseases: Specific IL receptor subunits (e.g. IL-1R1, IL-1R4 or IL-1R6) are blocked and binding of interleukins specific for one disease is inhibited. A new path was taken by Dr. Michael Wegmann (Research Center Borstel) and an international project group: Using the newly designed monoclonal antibody MAB-hR3 they blocked the central receptor subunit IL-1R3. In contrast to the other units, IL-1R3 is involved in signalling of all six pro-inflammatory interleukins of the IL-1 family (see figure, in green). The scientists posed the question, if this leads to a larger anti-inflammatory effect than blocking just one specific subunit.
The antibody MAB-hR3 was tested in three disease models in mice: peritonitis, psoriasis and allergic asthma. In each of these diseases, a distinctive interleukin profile leads to a pathological inflammation resulting in disease symptoms. Actually, the treatment with MAB-hR3 ameliorated symptoms in all three cases. Partially, the antibody was more effective than blocking the specific IL-1 receptor subunits. The scientists from Borstel found in their allergic asthma model that less immune cells were recruited to the lung and less mucus had been produced after treatment with the antibody. Moreover, the intensity of experimentally induced asthma attacks was reduced.
The researchers conclude that turning off the complete IL-1 receptor signalling leads to a broader anti-inflammatory effect, because it not just suspends binding of one or two, but of all six interleukins. This strategy might also reduce co-morbidities and exclude compensation of a missing interleukin by another. However, it might also suppress the immune system in a way that patients suffer from more frequent infections. „Indeed, we have to find out, if there are adverse side effects after blocking such a central molecule of the immune defense “, says Michael Wegmann, Head of the Group Asthma Exacerbation and Regulation in Borstel. „Nevertheless we were able to show that our antibody has a large therapeutic potential in a number of IL-1 receptor dependent diseases like asthma“.
For this project, scientists from the US, the Netherlands and Denmark worked together with DZL researchers from Borstel. They published their results in the latest edition of Nature Immunology.
Further information: Højen JF, Kristensen MLV, McKee AS, Wade MT, Azam T, Lunding LP, de Graaf DM, Swartzwelter BJ, Wegmann M, Tolstrup M, Beckman K, Fujita M, Fischer S, Dinarello CA (2019) IL-1R3 blockade broadly attenuates the functions of six members of the IL-1 family, revealing their contribution to models of disease. Nat Immunol 20: 1138-1149
/jbul
Calendar
Upcoming events
2024-04-30
Virtual DZL AID Kick-off Seminar
2024-05-29
74th Meeting of ARCN
2024-06-05
